03 Mar Art of Re-entry: Practical Strategies for When Your Antegrade Wire Is Subintimal
Re-entering the true lumen after an antegrade wire has gone subintimal is one of the most challenging parts of chronic total occlusion work. It is equal parts planning, technical skill, and adaptability. This guide walks through practical options, preparation, device use, and troubleshooting to help you choose the right strategy and improve the odds of a successful re-entry.
Table of Contents
- Why re-entry planning matters
- Quick overview: Re-entry options
- Common pitfall: Lost vessel clarity after parallel wire or prolonged attempts
- Step 1: Plan re-entry before you lose options
- Tools and techniques
- Case examples and learning points
- Troubleshooting common problems
- Decision checklist before attempting re-entry
- Practical tips from the lab
- Conclusion
- Frequently asked questions
- Further reading and resources
Why re-entry planning matters
When a wire enters the subintimal space, your choices narrow quickly. The initial decisions you make — whether to redirect, escalate, use a parallel wire, attempt a dedicated re-entry device, try tip detection with intravascular ultrasound, or go retrograde — shape the anatomy you will be dealing with minutes later.
A key principle: the first three options (redirecting the same wire, escalating the wire, or using a parallel wire with escalation) are mutually exclusive in practical terms. If you stick with one technique for too long, the vessel clarity degrades and other techniques become less likely to work.
Quick overview: Re-entry options
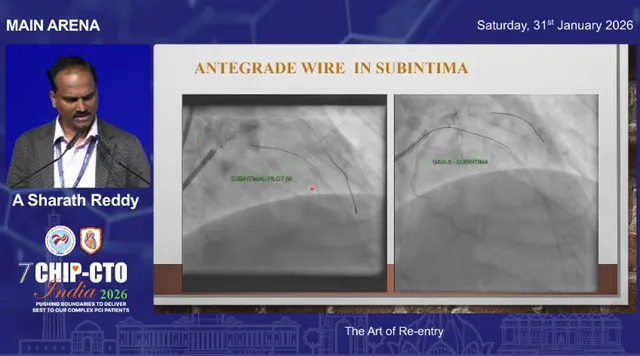
- Redirect same wire — try to recross into true lumen without exchanging or changing position.
- Wire escalation — use stiffer or different polymer wires to penetrate back into true lumen.
- Parallel wire with escalation — create a new channel beside the first wire using a stiffer wire.
- Dedicated re-entry device (Stingray) — intentionally enter subintimal space and use Stingray system to exit into true lumen.
- Tip detection ADR using IVUS (Iris/IVUS-guided) — visualize the vessel from within the subintimal space and guide the wire back into lumen.
- Retrograde approach — approach the CTO from collaterals to facilitate re-entry, sometimes with balloon protection (facilitated ADR).
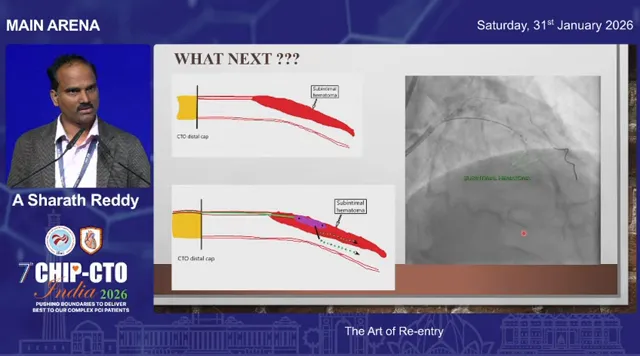
Common pitfall: Lost vessel clarity after parallel wire or prolonged attempts
When you attempt redirection or parallel wire techniques, repeated manipulation and time cause the vessel clarity to degrade. Contrast tracks obscure the lumen and a growing subintimal hematoma compresses the true lumen. When that happens, even a device like Stingray may have a much lower chance of success.
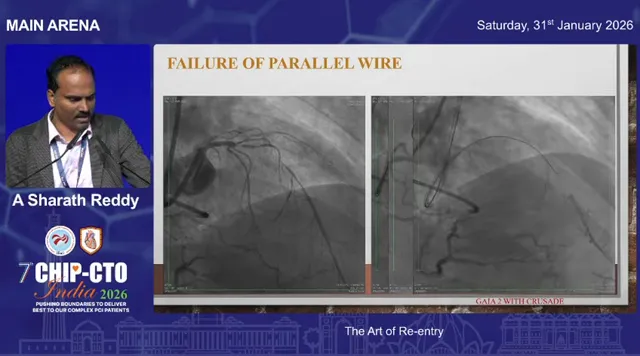
The practical takeaway is to avoid prolonged trial-and-error with multiple techniques in sequence. Decide early, keep your re-entry zone protected, and switch tactics before the vessel becomes unusable.
Step 1: Plan re-entry before you lose options
For any case without a reliable interventional collateral, plan your re-entry zone up front. CT angiography is exceptionally helpful for mapping the distal lumen and identifying calcium-free windows suitable for re-entry. If you anticipate no retrograde options, obtain a CT and choose a re-entry spot you can protect from knuckle tracks and subintimal hematoma.
Ask yourself:
- Where is the planned site of re-entry (choose the most calcium-free and visible segment)?
- What support will you have for device delivery?
- What is your dissection or crossing strategy — will you deliberately enter subintima or was it accidental?
- How will you save your re-entry zone from intramural hematoma?
- What re-entry method will you choose?
Tools and techniques
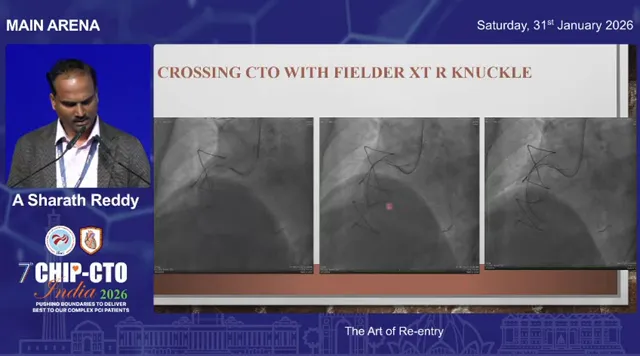
Knuckle and gladius strategy
A sliding “gladius” knuckle is often used to traverse long segments safely in the subintimal plane. Unlike a rolling knuckle, a sliding knuckle moves forward smoothly and can sometimes, unpredictably, re-enter the true lumen on its own. Always confirm position using a workhorse wire into nearby branches to verify entry into true lumen.

Stingray re-entry device
The Stingray system is the go-to when you intentionally perform antegrade dissection and re-entry (ADR) or when the wire has gone subintimal but you have a clear re-entry territory.
Practical steps when using Stingray:
- Choose the re-entry zone where the vessel is visible angiographically and not heavily calcified.
- Enhance support: place an anchor or support wire in a side branch or distal vessel to facilitate device delivery.
- Create a space for the Stingray: dilate with a 1.0 mm balloon, advance a Corsair or dedicated microcatheter, or exchange to a stiff support wire to allow the Stingray balloon to track.
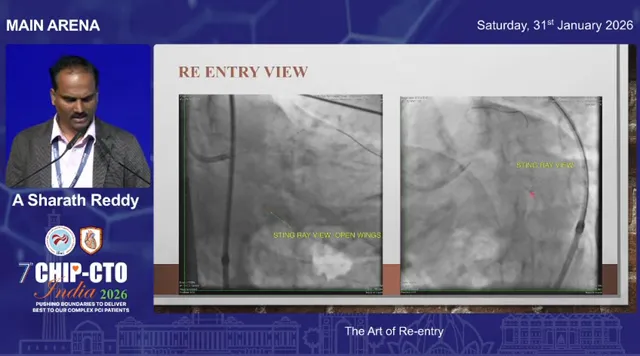
- Proper imaging orientation is essential. Recognize the “not-face” or side view versus the correct en face view needed for re-entry. Inject contrast through the Stingray balloon to determine lumen relation.
- Use trial-and-error exits: any exit port can orient toward true lumen. Try a Stingray wire exit, then swap to a workhorse or Pilot wire once you confirm true lumen access.
IVUS-guided tip detection (Iris catheter)
When tactile or angiographic clues are insufficient, intravascular ultrasound gives direct visualization of the device tip relative to the true lumen. IVUS-guided tip detection allows you to position a wire precisely under imaging guidance to re-enter the lumen.
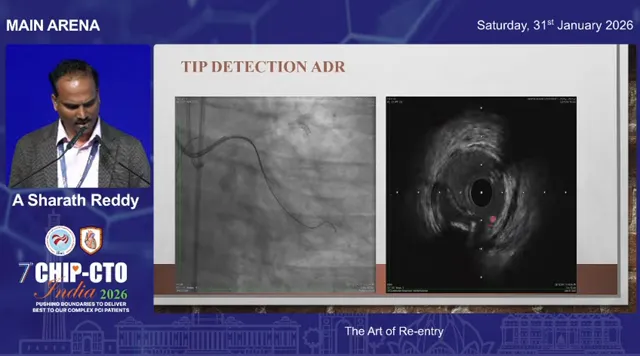
This is particularly useful when the subintimal space is large and the distal vessel morphology is preserved. In some cases, if the subintimal space is massive, a conservative strategy may be to perform balloon-only treatment of the created channel and stage further intervention later.
Facilitated ADR with retrograde balloon protection
In patients with poor or absent collaterals and prior CABG or occluded SVGs, facilitated ADR can be lifesaving. The technique uses a retrograde wire and balloon to protect the true lumen while an antegrade wire is directed toward the balloon for a controlled re-entry.
- Go retrograde to place a wire into the distal true lumen (for example PDA).
- Advance a balloon over that retrograde wire and inflate to protect the lumen.
- Direct your antegrade wire (Gaia 2/3 or similar) toward the balloon; once the antegrade wire exits the subintimal space, it will meet the protected true lumen.
Facilitated ADR effectively protects side branches and distal circulation, and it allows more aggressive knuckling proximally without jeopardizing the distal lumen.
Case examples and learning points
Case: Accidental knuckle re-entry
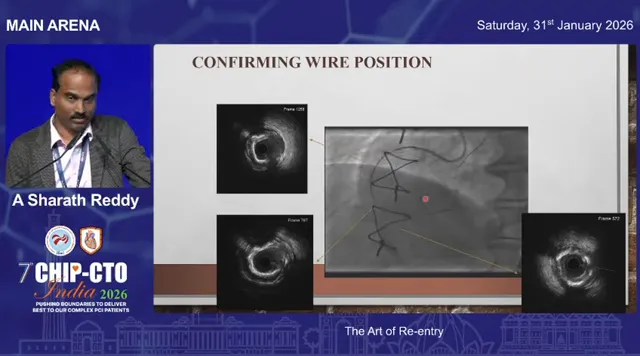
A gladius knuckle descended smoothly and unexpectedly re-entered true lumen. Confirmation came from a workhorse wire advancing into side branches and IVUS appearance consistent with true lumen placement. Lesson: sometimes the anatomy cooperates; always verify with branch wiring or IVUS before committing.
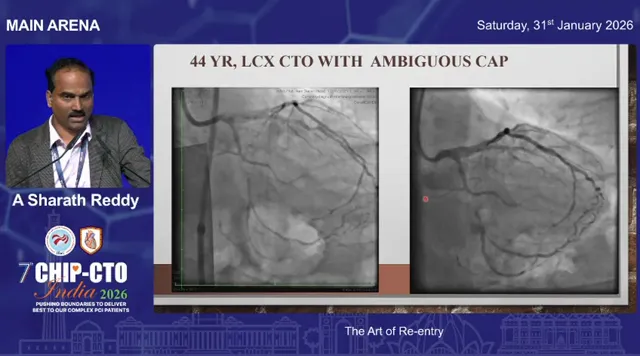
Case: Ambiguous cap LCx CTO managed with Stingray
After a proximal cap puncture and initial wiring ended up subintimal, the operator chose a clear angiographic re-entry zone. A support wire was placed in a side branch, the microcatheter was advanced and dilated to create space, then a Stingray was advanced over a stiff support wire. By obtaining the correct imaging perspective and injecting contrast through the Stingray balloon, the lumen relationship became obvious and the Stingray wire was used to exit into true lumen. Swap to a Pilot wire and complete the case while preserving side branches.
Case: Difficult re-entry with increased subintimal hematoma
In a left main CTO and distal disease scenario, repeated attempts with pilot or Gaia wires created a large subintimal hematoma that compressed the true lumen. Initial Stingray attempts produced buckling of the swap wire, indicating persistent subintimal position. The solution was to decompress the subintimal hematoma using STRAW (subintimal transcatheter withdrawal) maneuvers and to “bob-slide” the Stingray to identify a closer true lumen target. After reducing the hematoma and repositioning, successful re-entry was achieved.
Troubleshooting common problems
When the swap wire keeps buckling
Buckling of the swap wire is a classic sign you are still in the subintimal space. Options:
- Reduce subintimal hematoma with STRAW or aspiration techniques.
- Move the Stingray slightly (bob-sliding) to search for a point where the true lumen comes closer.
- Consider IVUS-guided tip detection if repositioning fails.
When vessel is heavily calcified
Calcified CTO segments are poor candidates for blind re-entry. Use pre-procedural CT to pick a calcium-free window; if none exists, consider a retrograde strategy or prepare for a more complex plan including possible hybrid surgical options.
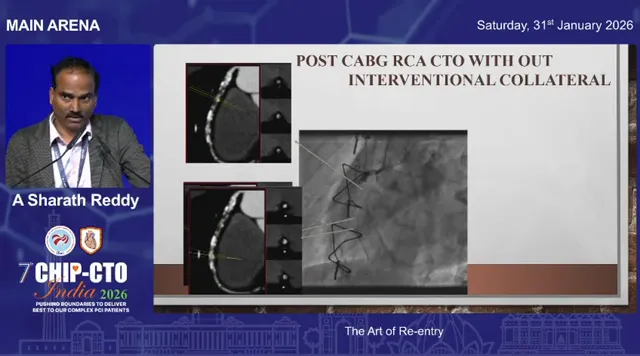
No interventional collateral and failed re-entry
In cases with no suitable retrograde route and failed antegrade re-entry despite best efforts, an “investment” strategy can be chosen. Create a controlled subintimal channel, treat it with balloon angioplasty if necessary, and stage further treatment later. IVUS can confirm an entry point from the false lumen to the true lumen that preserves distal perfusion and facilitates later reattempts.
Decision checklist before attempting re-entry
- Is there an interventional collateral? If yes, consider retrograde or facilitated strategies.
- Is the selected re-entry zone free of heavy calcium on CT or angiography?
- Can you enhance support (anchor wire, trap balloon) to deliver re-entry devices?
- Do you have equipment available: microcatheters, Corsair, Stingray balloon, specialized wires (Gaia 2/3, Pilot), IVUS/Iris?
- Do you have a plan to reduce subintimal hematoma if it develops?
- If re-entry fails, what is your bail-out? (Investment strategy, staged approach, referral for alternative therapy)
Practical tips from the lab
- Protect your re-entry zone: avoid passing knuckles through the selected re-entry window; use CT to pre-identify safe spots.
- Use small balloon dilation (1.0 mm) or microcatheter advancement to create a pathway for Stingray rather than forceful bodywork.
- Get the correct Stingray view: recognizing face versus side views and injecting through the Stingray improves localization.
- Keep option flexibility: if a technique is failing and time is increasing, switch early to an alternative rather than persisting and compromising the vessel.
- Use facilitated ADR when safe retrograde access exists — balloon protection preserves true lumen and side branches.
- IVUS is invaluable for tip detection, assessing whether the swap wire is truly in the lumen, and for planning staged interventions.
Conclusion
Re-entry is an art informed by planning, imaging, and the timely selection of techniques. Choosing the right strategy early, protecting the planned re-entry zone, and being prepared to switch when one approach fails are the practical keys to success. Use CT for planning, enhance support for device delivery, and remember that tools such as Stingray, IVUS tip detection, and facilitated retrograde balloon protection each have specific roles depending on the anatomy and collateral status.
When a case becomes hostile — large subintimal hematoma, diffuse disease with no clear re-entry window — an investment and staged approach is a valid and safe alternative to aggressive, high-risk attempts that compromise the distal vessel.
Frequently asked questions
What are my first-line choices when my antegrade wire goes subintimal?
First-line options are: attempt redirection of the same wire, escalate to a stiffer wire, or use a parallel wire with escalation. If those are not feasible or fail, consider dedicated re-entry with Stingray, IVUS-guided tip detection, or retrograde facilitated ADR depending on collateral availability and vessel anatomy.
How does Stingray re-entry work and when should I use it?
Stingray is a balloon-based system that intentionally positions an exit port near the true lumen. After creating space and obtaining the correct imaging orientation, a dedicated Stingray wire is pushed through the appropriate exit port to access the lumen. Use Stingray when you have a clear re-entry zone that is not heavily calcified and you can provide adequate support for device delivery.
What signs tell me the swap wire is still subintimal?
Buckling of the swap or workhorse wire when attempting to advance is a classic sign of persistent subintimal positioning. Lack of forward progress with wire exchanges despite Stingray attempts suggests hematoma or distance between Stingray and true lumen. Consider STRAW maneuvers, repositioning, or IVUS guidance.
When should I consider facilitated ADR with a retrograde balloon?
Facilitated ADR is indicated when there is usable retrograde access and you want to protect the true lumen while advancing an antegrade wire. It is especially useful in post-CABG patients or when collaterals allow placement of a balloon in the distal lumen to serve as a target.
Is it ever acceptable to stop and adopt an investment strategy?
Yes. When re-entry attempts threaten the distal vessel, or the disease is diffuse with no realistic re-entry target, creating a controlled subintimal channel, treating it conservatively with ballooning, and staging a later attempt can preserve distal perfusion and reduce procedural risk.
Further reading and resources
For ongoing skill development, study CT planning for CTOs, Stingray technique tutorials, IVUS-guided ADR cases, and algorithms for hybrid CTO approaches. Practical experience, deliberate case selection, and team coordination remain central to mastering the art of re-entry.
More Frequently Asked Questions (FAQs)
Art of Re-entry: Practical Strategies for When Your Antegrade Wire Is Subintimal
1. What does it mean when the antegrade wire is subintimal?
It means the guidewire has left the true lumen of the coronary artery and entered the subintimal (false lumen) space between the intima and media. This is common during chronic total occlusion (CTO) interventions and does not automatically mean failure — but it does change your strategy immediately.
2. Is going subintimal always a mistake?
No.
Subintimal tracking may be:
- Accidental (during wire escalation or cap penetration)
- Intentional (as part of Antegrade Dissection and Re-entry, ADR)
In modern CTO practice, controlled subintimal navigation is often deliberate — especially when using devices like Stingray Re-Entry System.
The key question is not “Did I go subintimal?” but rather “Do I have a viable re-entry plan?”
3. What are my first-line options after entering the subintimal space?
Your immediate options include:
- Redirecting the same wire
- Escalating to a stiffer or different wire
- Using a parallel wire technique
- Switching early to a dedicated re-entry device
- Considering retrograde access (if available)
Important principle:
Prolonged attempts with multiple techniques reduce vessel clarity and increase subintimal hematoma formation.
4. How do I decide between parallel wire and Stingray?
Use a parallel wire strategy when:
- The distal lumen is clearly visible
- Hematoma is minimal
- The subintimal space is not excessively expanded
Use Stingray when:
- You have a clear angiographic re-entry zone
- You intentionally created a subintimal plane (ADR)
- Parallel attempts have failed but vessel clarity is preserved
- You can deliver the device with adequate support
Early decision-making preserves your re-entry window.
5. What is the biggest mistake operators make during re-entry?
The most common error is waiting too long to switch strategies.
Repeated manipulation:
- Increases contrast staining
- Expands intramural hematoma
- Compresses the distal true lumen
- Reduces Stingray success
Once vessel clarity degrades, even excellent technique may fail.
6. How do I recognize that my swap wire is still subintimal?
Classic signs include:
- Wire buckling during advancement
- Lack of distal branch wiring
- No side branch filling
- Persistent resistance despite device attempts
Buckling is especially common after Stingray exit attempts when the exit point is still facing false lumen.
7. How can I reduce subintimal hematoma?
Options include:
- STRAW (Subintimal Transcatheter Withdrawal technique)
- Aspiration maneuvers
- Gentle decompression
- Moving the re-entry zone proximally or distally
- IVUS-guided targeting of a closer lumen segment
Reducing hematoma can dramatically improve re-entry success.
8. When should I use IVUS-guided tip detection?
Intravascular Ultrasound (IVUS) tip detection is particularly helpful when:
- Angiographic cues are unclear
- Subintimal space is large
- The relationship between wire and lumen is uncertain
- Stingray attempts are inconclusive
IVUS provides direct visualization of:
- True lumen location
- Distance from wire tip
- Hematoma size
- Calcium distribution
It reduces guesswork and improves precision.
9. What is facilitated ADR and when is it useful?
Facilitated ADR involves:
- Retrograde wire placement in distal true lumen
- Balloon inflation to protect the lumen
- Directing antegrade wire toward the balloon
It is especially useful:
- In post-CABG patients
- When usable collaterals exist
- When protecting distal side branches is critical
This hybrid method increases safety and re-entry accuracy.
10. Should I always try retrograde if antegrade re-entry fails?
Not necessarily.
Consider retrograde when:
- Interventional collaterals are suitable
- Operator expertise allows safe crossing
- The distal vessel is large and worth preserving
Avoid retrograde if:
- Collaterals are high risk
- Patient instability increases procedural risk
- Time and contrast load are excessive
Strategy must balance benefit versus risk.
11. What is the “investment strategy” in CTO re-entry?
An investment strategy means:
- Creating a controlled subintimal channel
- Preserving distal perfusion
- Possibly ballooning gently
- Staging the definitive procedure later
It is appropriate when:
- Re-entry threatens the distal vessel
- Hematoma is large
- No realistic re-entry target remains
- Procedural risk is escalating
Stopping early is sometimes the safest choice.
12. How important is CT planning before CTO re-entry?
Pre-procedural CT angiography is extremely valuable for:
- Identifying calcium-free re-entry zones
- Mapping distal vessel size
- Planning landing zones
- Predicting difficulty
When no retrograde option exists, CT planning becomes even more critical.
13. What wires are commonly used for re-entry?
Depending on strategy, operators may use:
- Gaia 2 / Gaia 3
- Pilot series
- Workhorse wires for confirmation
- Dedicated Stingray wire
Selection depends on:
- Vessel morphology
- Distance to lumen
- Calcium burden
- Support level
Wire choice should match anatomical need — not habit.
14. How do I protect my re-entry zone?
- Pre-identify it before knuckling
- Avoid sending large knuckles through it
- Limit prolonged manipulation
- Enhance guide support
- Consider early device use rather than repeated blind attempts
Protecting the re-entry zone improves procedural success significantly.
15. What imaging view is critical when using Stingray?
Proper orientation is everything.
Recognize:
- “Side view” (incorrect alignment)
- “En face” view (correct alignment)
- Contrast injection through balloon to define lumen relationship
Misinterpretation of imaging orientation is a common cause of failure.
16. When should I stop trying?
You should strongly consider stopping when:
- Distal vessel is compromised
- Hematoma continues expanding
- Contrast load becomes unsafe
- Radiation time escalates
- No viable re-entry window remains
Knowing when to stage is part of mastering the art of re-entry.
17. What is the single most important principle in re-entry?
Decide early. Switch early. Protect the distal vessel.
Re-entry success depends less on force and more on:
- Planning
- Imaging
- Strategy selection
- Flexibility
Aggressive persistence without strategy adaptation often leads to vessel loss.
18. Can accidental knuckle re-entry happen?
Yes — occasionally a sliding knuckle may re-enter spontaneously.
However, always confirm true lumen position with:
- Side branch wiring
- Distal contrast filling
- IVUS confirmation
Never assume re-entry without proof.
19. Is heavy calcification a contraindication for Stingray?
Heavily calcified segments are poor candidates for blind re-entry.
In such cases:
- Use CT to locate a calcium-free window
- Consider IVUS guidance
- Evaluate retrograde options
- Prepare for advanced plaque modification strategies
Calcification dramatically reduces exit success.
20. What separates expert operators from beginners in CTO re-entry?
Experts:
- Plan re-entry before crossing
- Preserve vessel clarity
- Switch techniques decisively
- Use imaging liberally
- Accept staging when appropriate
Mastery is not about forcing success — it is about choosing the safest path to it.


No Comments